Ionic Bonds in Ceramic Material
The binding energy for ionic bonding varies from 150-370
kcal/mol, the same for metallic bond varies from 25-200 kcal/mol.
The stiffness of a bond is related to the binding energy by the
relationship S = (d2U / dr2), while the force between the atoms
could be derived from the binding energy by evaluating
F= (dU / dr), where the binding energy for an ionic bond is defined
by the relationship:
U= U0 – q2 / (4π ε0 r) + B/rn
Binding energy for a relatively weaker bond like Van der Walls
bonding is defined as:
U = - A/r6 + B/r12
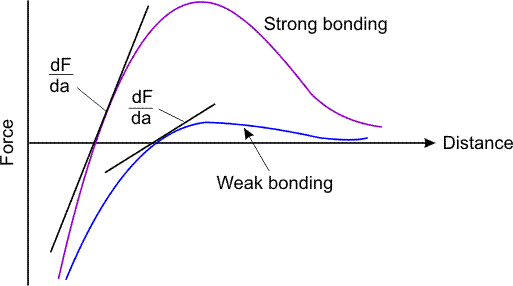
If you calculate the Force-deflection relationship for the two type of
bonding you will get the figure below which clearly shows why
ionic bonding has steeper force deflection relationship implying
higher modulus of elasticity.
|