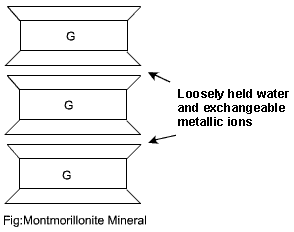
Montmorillonite and illite clay minerals are the most common. A basic three-layer sheet unit is formed by keeping one silica sheet each on the top and at the bottom of a gibbsite sheet. These units are stacked to form a lattice as shown.
Montmorillonite Mineral
The bonding between the three-layer units is by van der Waals forces. This bonding is very weak and water can enter easily. Thus, this mineral can imbibe a large quantity of water causing swelling. During dry weather, there will be shrinkage.
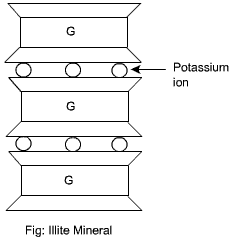
Illite Mineral
Illite consists of the basic montmorillonite units but are bonded by secondary valence forces and potassium ions, as shown. There is about 20% replacement of aluminium with silicon in the gibbsite sheet due to isomorphous substitution. This mineral is very stable and does not swell or shrink.