Print this page
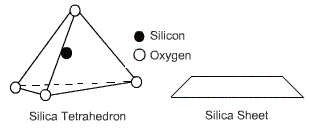
A tetrahedral unit consists of a central silicon atom that is surrounded by four oxygen atoms located at the corners of a tetrahedron. A combination of tetrahedrons forms a silica sheet.
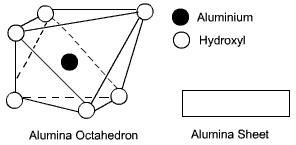
An octahedral unit consists of a central ion, either aluminium or magnesium, that is surrounded by six hydroxyl ions located at the corners of an octahedron. A combination of aluminium-hydroxyl octahedrons forms a gibbsite sheet, whereas a combination of magnesium-hydroxyl octahedrons forms a brucite sheet.