|
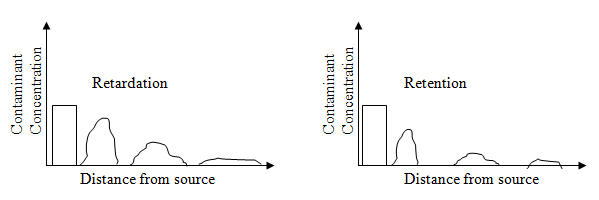
Fig. 2.14 Attenuation process due to soil-contaminant interaction |
For an effective waste management, retention process is more ideal than retardation. For proper prediction of contaminant fate, it is very essential to know whether the contaminant is retained or retarded. The important reactions determining attenuation are discussed as follows:
1) Hydrolysis
Hydrolysis is the reaction of H+ and OH- ions of water with the solutes and elements present in the pore water. Such a reaction would continue only if the reaction products are removed from the system. Water is amphiprotic in nature (Yong 2001), which means it can act as acid or base. According to Bronsted-Lowry concept an acid is a proton donor and base is a proton acceptor. According to Lewis, acid is an electron acceptor and base is an electron donor. As discussed earlier, soil minerals have ionized cations and anions (metal ions) attached to it that results in a particular pH level in soil-water system. Hydrolysis reaction of metal ions can be represented as
| MX + H2O → MOH + H+ + X- | 2.39 |
The reaction increases with decrease in pH, redox potential and organic content and increases with temperature. Hydrolysis can be an important reaction in the process of biodegradation. For example,
| (R-X) + H2O → (R-OH) + X- + H+ | 2.40 |
where R is an organic molecule and X is halogen, carbon, nitrogen or phosphorus. R-X is resistant to biodegradation. The reaction introduces OH in place of X making organic molecule susceptible to biodegradation.
2) Oxidation-reduction (redox) reaction
Oxidation-reduction (redox) reaction involves transfer of electrons between the reactants. In general, transfer of electrons is followed by the proton transfer also. Soil pore water provides medium for oxidation-reduction reaction which can be biotic and/or abiotic. Microorganisms present in the soil utilize oxidation-reduction (redox) reactions as a means to derive energy required for its growth. Hence, these microorganisms act as catalysts for reactions (redox) involving molecular oxygen, soil organic matter and organic chemicals. For inorganic solutes, redox reaction results in the decrease or increase in the oxidation state of the atom. This is important because some ions have multiple oxidation states and hence would influence the soil-contaminant interaction. It is found that biotic redox reactions are more significant than abiotic redox reaction.
The redox potential Eh represented by Eq. 2.41 determines the possibility of oxidation-reduction reaction in the soil-contaminant system.
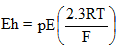 |
2.41 |
E is the electrode potential, pE represent negative logarithm of electron activity e-, R is the gas constant, T is the absolute temperature, and F is the Faraday constant. At a temperature of 250C, Eh = 0.0591pE. Factors affecting Eh include pH, oxygen content or activity, and soil water content.
3) Complexation
Complexation is the reaction between metallic cations and anions called ligands. The inorganic ligands such as Cl-, B-, F-, SO4-2, PO4-3, CO3-2 and organic ligands such as amino acids take part in
complexation reaction. For example,
Mn+2 + Cl- → MnCl+
Complexation can also occur in series, such that complex formed from one reaction can react with another ligand.
An example of such a reaction taken from Reddi and Inyang 2000 is presented
below.
Cr+3 + OH- → Cr(OH)+2
Cr(OH)+2 + OH- → Cr(OH)2+
Cr(OH)2+ + OH- → Cr(OH)30
This indicates that the concentration of metals in the form of complexes also needs to be taken into account in addition to the free metal ion concentration. Else, the concentration of the metal transported downstream would always be more than the predicted concentration of the metal ion.