Cytochrome c oxidase:
Cytochrome c oxidase is an electron transfer protein found at the terminal position in the respiratory transport chain of animals, plants, aerobic yeasts and in some bacteria. It reduces molecular oxygen (O2) to H2O.
![]()
Active site:
The active site (where the reaction takes place) of cytochrome c oxidase is composed of a heme a3 and CuB which are ~ 4.5 Å apart from each other.
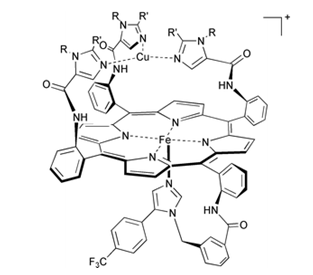
Figure 7.8. The active site of cyrochrome c oxidase. Pictute is taken from Annu. Rep. Prog. Chem., Sect. A: Inorg. Chem. , 2003, 99 , 565-588.
Proposed mechanism:
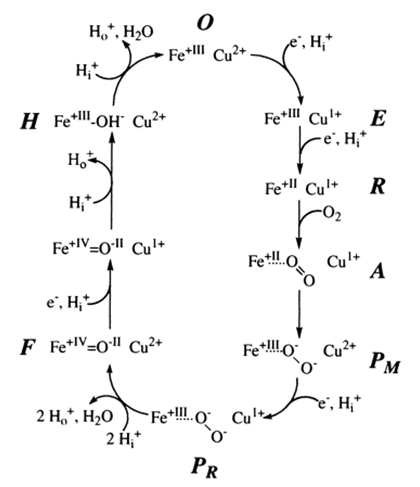
Figure 7.9. Proposed mechanism for the conversion of O2 to H2O by cytochrome c oxidase. Taken from PNAS October 27, 1998 vol. 95 no. 22 12819-12824. R = reduced form, O = oxidized form, P = peroxo form, H = hydroxyl form, F = ferryl form.
Cytochrome P450:
Heme cytochrome P450 is a large and diverse group of enzymes. The function of most cytochrome P450 enzymes is to catalyze the oxidation of organic substances.
The most common reaction catalyzed by cytochromes P450 is a monooxygenase reaction, e.g., insertion of one atom of oxygen into an organic substrate (RH) while the other oxygen atom is reduced to water:
![]()
Active site:
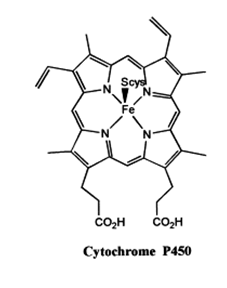
In the active site of cytochrome P450 iron is in +III oxidation state and it is in square pyramidal geometry where the basal plane is composed of four N atoms from the prophyrin ring and the fifth, i . e . the axial position is occupied by a cystine moiety. The name P450 arises from the position of the absorption band of the oxoform of the enzyme, i . e . oxo form shows absorption band at 450 nm in the UV-Vis spectrum.