Lux-Flood Definition (1939):
According to this theory, acid and base is define as, acid is an oxide acceptor and base is an oxide donor.
Example:
Here, CaO donates oxygen, therefore, it is a basic oxide. SiO 2 is an oxide acceptor, hence, it is a acidic oxide.
Merits and demerits:
Merit:
This approach emphasizes the acid and basic-anhydride aspects. Acidic oxides are acid anhydride and in the aqueous medium generate protonic acids, while, basic oxides are basic anhydride and generate hydroxyl ion in the aqueous medium.
Demerit:
The usefulness of this concept is limited to inorganic metal oxide and their reactions in molten state.
The Usanovich Definition (1939):
The Usanovich definition includes all reactions of Lewis acids and bases and extended the latter concept by removing the restriction that the donation or acceptance of electrons be as shared pairs. According to him,
an acid is any chemical species which (i) reacts with a base, or (ii) accept anions or electrons, or (iii) furnish cations; and a base is any chemical species which (i) reacts with acids, or (ii) gives up anions or electrons, or (iii) combines with cations.
Examples:
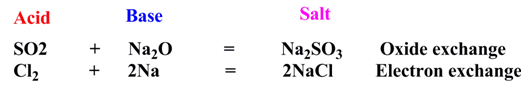
Merits and demerits:
Merits:
Lewis concept and protonic concepts have been covered under the Usanovis definition for acid-base. Besides that it states that the donation or acceptance of electrons need not take place as shared pairs. Accordingly, oxidation-reduction reactions may be classified as acid-base reaction.
Demerits:
This concept is extremely general and therefore, almost all reactions can be considered as acid-base reaction. Because of this, the convenience of treating a particular type of compounds as acids or bases has been lost.