Isomerism (geometrical and optical):

Some terms & their definitions:
Enantiomer : Stereoisomers which are not superimposable on their mirror images are called enantiomers.
Diastereoisomers : Stereoisomers which do not possess mirror image relation are called diastereoisomers.
Asymmetric molecule : A molecule without any symmetry (except c1) is classified as an Asymmetric molecule.
Dissymmetric molecule :
Geometric Isomerism: Geometric Isomers differ in the spatial arrangement of atoms within the same structural framework. It is also called cis- trans- isomerism.
Geometric Isomerism for coordination no. 4
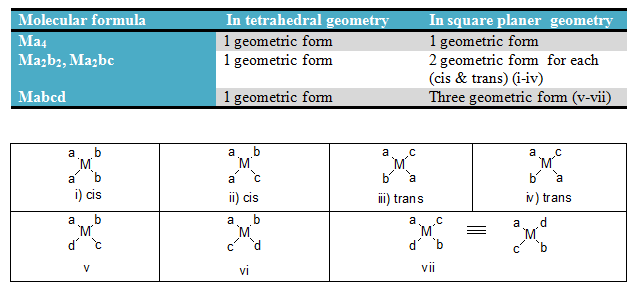
Q. How many geometrical isomer is possible for [PtCl2(NH3)2] ?
A) 1 B) 2 C) 3 D) 4
Answer: ( B )
Referance/Hints: It is Ma2b2 type molecule.
Q. How many geometrical isomer is possible for [Pt(NO2)(NH3)(py)(NH2OH)]NO2 ?
A) 1 B) 2 C) 3 D) 4
Answer: ( 3 )
Referance/Hints: It is Mabcd type molecule.