What is a matter?
Matter is something that occupies space, has mass, exists as solid, liquid or gas, and can be perceived by one or more senses.
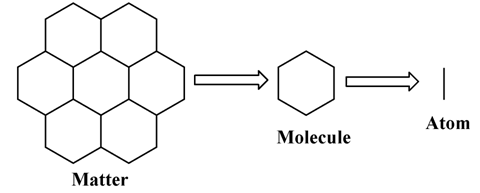
Figure 1.1. Composition of a matter. An arbitrary picture.
A matter can be taken as aggregation of molecules. Molecule is composed of atom(s).

Figure 1.2. Different states of a matter. Circle indicates a unit volume.
A matter can exist in three different phases.
- Solid phase: In this phase molecules are tightly bound to each other. High energy is required to separate molecules.
- Liquid phase: In this phase molecules are moderately bound to each other. Generally, moderate energy is required to separate constituent molecules.
- Gas Phase: In this phase intermolecular bonding is very weak. Molecules are far apart to each other and can be separated easily.
The Atomic model:
(A) Rurherford´s model (1911):

Figure 1.3. Scattering experiment of a - particle by gold layer.