All fuel cells have the same basic operating principle.
- An input fuel is catalytically reacted (electrons removed from the fuel elements) in the fuel cell to create an electric current.
|
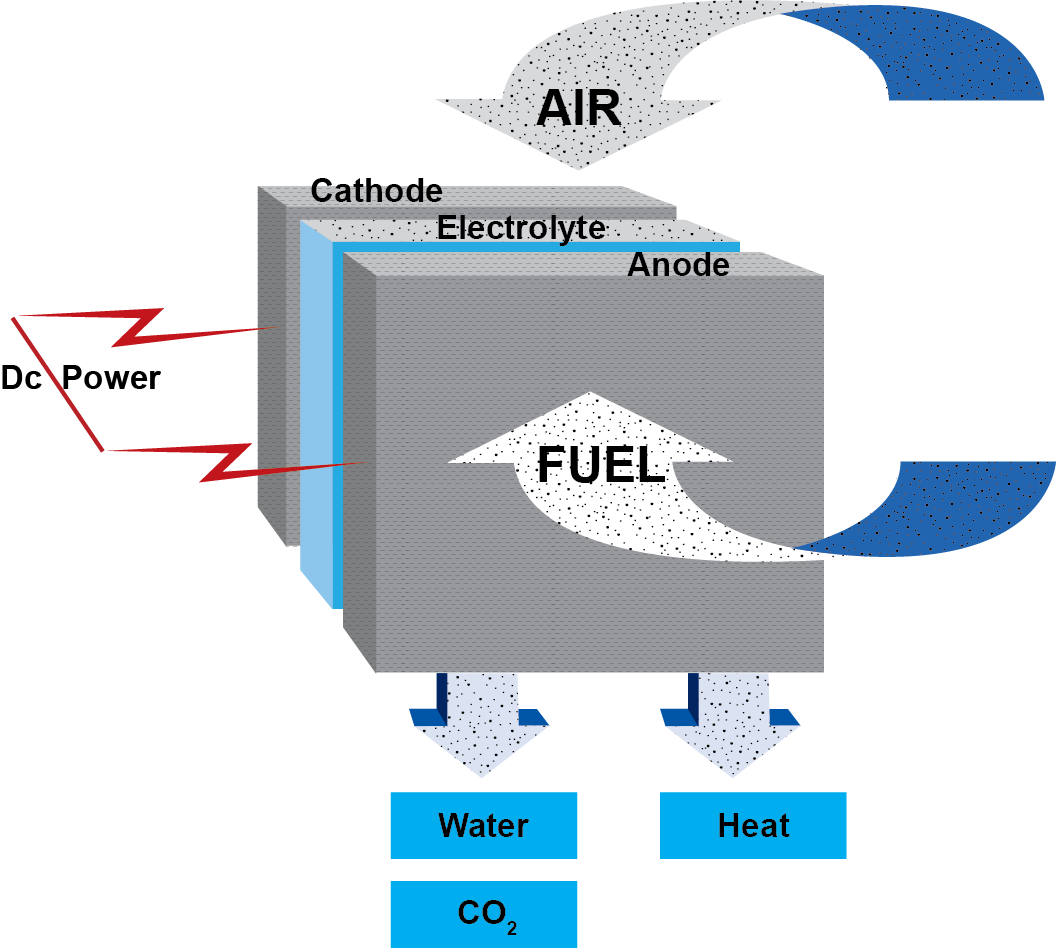 |
- Fuel cells consist of an electrolyte material which is sandwiched in between two thin electrodes (porous anode and cathode).
- The input fuel passes over the anode (and oxygen over the cathode) where it catalytically splits into ions and electrons.
- The electrons go through an external circuit to serve an electric load while the ions move through the electrolyte toward the oppositely charged electrode.
- At the electrode, ions combine to create by-products, primarily water and CO2. Depending on the input fuel and electrolyte, different chemical reactions will occur.
|

