Ultraviolet – Visible spectrophotometry
Introduction
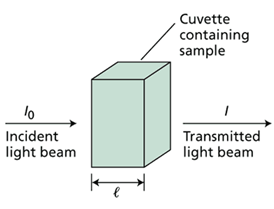
Many molecules absorb ultraviolet or visible light. The absorbance of a solution increases as attenuation of the beam increases. Absorbance is directly proportional to the path length, l, and the concentration, c, of the absorbing species. Beer's Law states that A = elc, where e is a constant of proportionality, called the absorbtivity.
|
Different molecules absorb radiation of different wavelengths. An absorption spectrum will show a number of absorption bands corresponding to structural groups within the molecule. For example, the absorption that is observed in the UV region for the carbonyl group in acetone is of the same wavelength as the absorption from the carbonyl group in diethyl ketone.
|