Formation of Microemulsions*
![]()
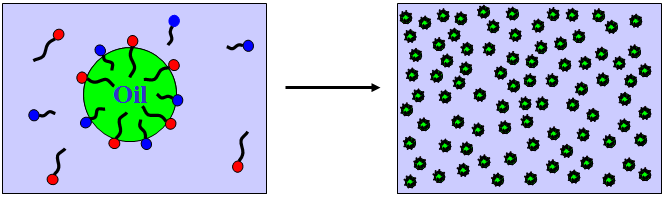
ΔGm = free energy change for microemulsion formation
ΔG1 = free energy change due to increase in total surface area
ΔG2 = free energy change due to interaction between droplets
ΔG3 = free energy change due to adsorption of surfactant at the
oil/water interface from bulk oil or water
ΔS = increase in entropy due to dispersion of oil as droplets