C60 fullerene has 60 pi electrons but a closed shell configuration requires 72 electrons. The fullerene is able to acquire the missing electrons by reaction with potassium to form first the K6C6−60 salt and then the K12C12−60 In this compound the bond length alternation observed in the parent molecule has vanished.
Synthesis of Fullerenes:
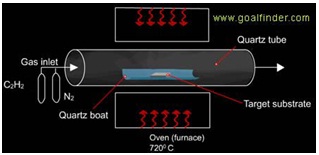
Chemical vapor deposition process of producing nanotubes uses a target substrate containing catalysts in a quartz tube; a hydrocarbon gas breaks apart and deposits carbon on the substrate. Through this method carbon nanotubes are produced using base growth mechanism.