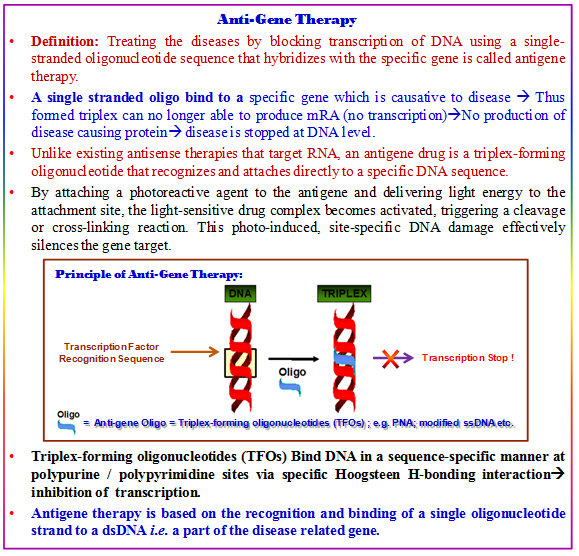
4.22.15. Antigene Gene Therapy
4.22.15.1. Introduction
Since the complete mapping of the human genome, there has been a considerable interest in exploring various strategies that would bring about specific gene silencing. Although siRNA technology has made an enormous step toward drug development which effectively shut down gene expression at the mRNA level (antisense therapy) but suffer from the problem of prolonged shut down of gene expression. Thus, treatment targeting at DNA level (anti-gene therapy) have come up to cure gene. Thus, a DNA-modifying agent has to so design as to bring about a site-specific DNA modification, as in the form of a double-strand binding or breaking, may result in a mutation that would knockout the gene’s function.
Antigene-based therapy require the targeting molecule to (a) enter the nucleus, (b) bind DNA, (c) stop transcription and/or (d) elicit specific DNA damage without hampering with normal cellular function. In 1987, Le Doan et al., and et al. were the first to show the potential of attaching a DNA-modifying agent to a triplex-forming oligonucleotide (TFO) to achieve site-specific DNA damage to double-stranded DNA (dsDNA).
Triplex-Forming Oligonucleotides (TFO): TFOs are defined as DNA/RNA or DNA/RNA analogs that have the propensity of forming a triplex with a given dsDNA target molecule. They are typically composed of a stretch of homopurines or homopyrimidines, a prerequisite that is usually required for efficient triplex formation. Ben Gaied et al., in 2009 showed that modified nucleobases or appended DNA intercalators that, when included into a TFO, allow the generation of stable triplex helices even with a dsDNA target. In addition, several DNA/RNA analogs have been shown to improve triplex formation, thus show their potency as antigene agents. Other TFO-enhancing elements include nucleotides such as locked nucleic acids.
Peptide nucleic acid (PNA): Peptide nucleic acid (PNA), introduced Nielsen in 1991, is a DNA mimic composed of a peptide like backbone. These DNA analogs have found many applications in the antigene field because of their high propensity to bind to duplex DNA via strand invasion. In a PNA-DNA-PNA triplex, one PNA strand forms Hoogsteen hydrogen bonds with the DNA target, whereas the other PNA invades the DNA duplex, forming Watson–Crick hydrogen bonds with the same DNA target while displacing the complementary DNA strand.
Photoactivated TFO conjugates: Photoactivated TFO conjugates are also found to be potential candidates for improving the antigene-based performance of oligonucleotides. By binding the target gene and damaging it is the basic working principle of such photoactivated TFO. As for example, psoralens (tricyclic compounds containing furan and pyron rings) are bifunctional photoreagents that can intercalate into double-stranded nucleic acids and upon UV irradiation undergo a (2+2) cycloaddition reaction with pyrimidine bases. PNA-Benzophenone upon photo irradiation at 330–400 nm form PNA-DNA crosslinks DNA-Pyrene conjugate at 355nm breakes dsDNA.