3.11.2.2. Hydrolases (esterase, lipase): Hydrolytic reactions → Catalyze bond cleavage by the introduction of water; Hydrolysis-formation of esters, amides, lactones, epoxides, nitriles, anhydrides:
Hydrolases:
- hydrolytic cleavage of C-O, C-N, C-C and some other bonds
- favourite class of enzymes for organic chemistry
- no cofactor needed
- large number of readily available enzymes
- reversible reaction (e.g., amide- or ester-synthesis)
- relative high stability under non-natural conditions
- often wide substrate range
Lipases:
- Among the biocatalysts in organic synthesis, lipases are the most frequently used. In particular, this class of enzyme is able to perform enantioselective hydrolytic reactions and catalyzes the formation of a wide range of ester and amide bonds.
- natural function - hydrolysis or re-esterification of triglycerides (oils and fats)
- Laundry detergents, modification of natural oils and fats etc.
- offers often excellent stereoselectivity - synthesis of optically pure compounds
- Lipases are highly active in organic solvents
- the most frequently used enzymes in organic synthesis
- Resolution of alcohols
- Synthesis of versatile building blocks in organic synthesis
- kinetic resolution by hydrolysis
- kinetic resolution by acylation - activated acyl donors or enol esters (e.g., vinyl or isopropenyl acetate) reaction irreversible as alcohol generated undergo keto-enol tautomerisation
Esterases:
- split esters into an acid and an alcohol
- only few applications for synthesis of optically pure compounds
- moderate enantioselectivity and limited commercial availability
Peptidases, Amidases and Acylases:
- formation and hydrolysis of amide links
- thermodynamically controlled synthesis - shift equilibrium towards synthesis by use of organic solvents, increase concentration of starting material or removing product.
- kinetically controlled synthesis - activation of carboxyl component usually by ester or amide
- Peptide synthesis → coupling of two amino acids
- advantages of enzyme-catalyzed peptide synthesis: mild conditions , high region and enantioselectivity, no racemisation
- large scale production of Aspartame - low-calorie sweetner (DSM, NutraSweet)
- production of Kyotorphin - analgesic dipeptide (Tyr-Arg)
- synthesis of β-lactam peptide antibiotics (penicillines and cephalosporines)
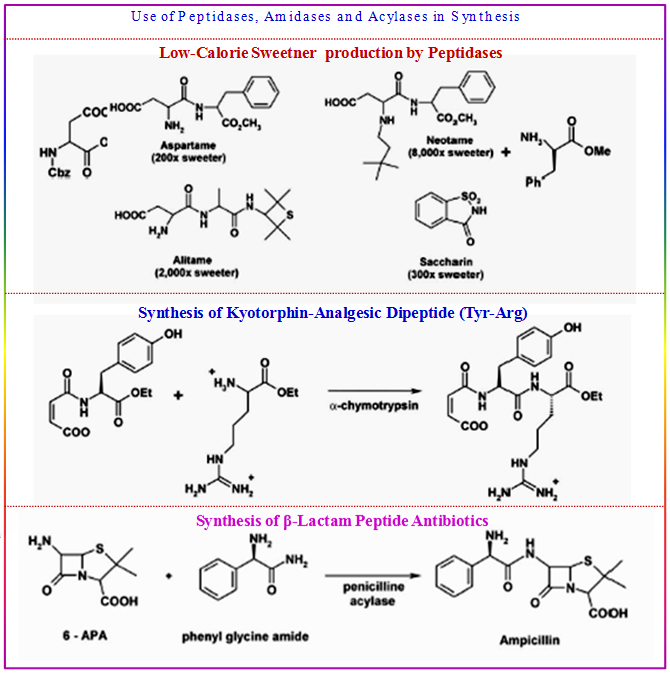
Figure 3.23: Examples of peptidases, amidases and acylases in organic reactions.