In each case we are observing the change in the number of aggregates with time, which tells us about the loss of primary particles from the system. A simple experiment for this would be to observe the concentration at which aggregation becomes apparent using tubes containing different electrolytes. This can also be quantified using the percentage transmission of the system as a measure of the number of particles in suspension. The measurement of rate of aggregation provides a more precise determination of ccc. At higher electrolyte concentrations the rate increases to the plateau value (as seen in Fig. 10.13) representing the fast or diffusion limited rate.
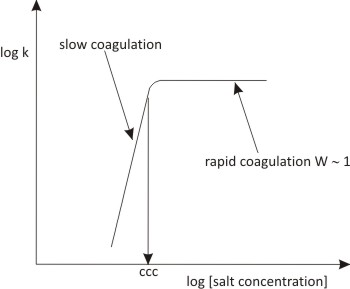
Fig. 10.13: Rate of coagulation against salt concentration (indicating c.c.c.)
Again, it should be noted that the analysis of the rate constant in terms of the diffusion of single particle is strictly the initial rate. As we progress into the coagulation process the particle number changes and the mechanism changes to one where the large much less mobile aggregates get larger by adding singlets and accurately describing the rates becomes quite complex.
|