As all the particles crossing the shell are coagulating and each collision results in coagulation, the coagulation rate is simply the collision rate. Thus it can be said that jA is the rate of coagulation per particles per unit time. If we consider all the particles, the rate at which particles are coagulating is given by
which has the characteristic form of a second-order process with a diffusion-controlled
rate.
In the above equation, Kr is the rate constant for rapid coagulation given by
Similar to molecular kinetics, we can express the rate coefficient in the form of an Arrhenius expression:
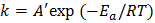
In our case the pre-exponential factor is simply the collision frequency. We have assumed that when particles collide they stick. There is no barrier in place and thus the activation energy is zero. Hence Kr, in this case is independent of temperature.
From Einstein-Stokes equation for the Brownian self-diffusion, diffusion coefficient for spherical particles is given by
Combining equation (10.19) and (10.20) we get
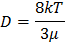
|