Kinetics of Coagulation
Equilibrium thermodynamics does not dictate the rate at which processes occur. Manycolloidal dispersions have kinetic stability, even though they are unstable thermodynamically. Therefore, the kinetics of coagulation is equally important for the study of stability of colloids.
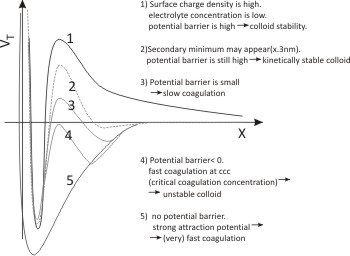
Fig. 10.7: Conditions of colloid stability
Energy between two colloidal particles can be represented by the above figure.When there is some energy barrier, (~ kT) the coagulation is called slow coagulation (case 3 in Fig. 10.7).In the absence of an energy barrier, the coagulation is rapid, limited only by the rate of diffusion towards each. Coagulation will occur between two particles when they are at the separation of global minimum of interaction energy. This is the condition when the coagulation is thermodynamically favorable. In this section we will discuss the study the kinetics of coagulation for both slow coagulation and rapid coagulation.
|