Effect of Concentration
We saw above that these interaction energies also depend upon the salt concentrations. At low salt concentrations (n0), the Electrostatic Repulsion term dominates and the overall interaction energy is positive. For high salt concentrations, the repulsive interactions have short-range effect and here Van-der Waals attractive term becomes dominant, leading to an overall Attractive Interaction.
Let us look the effect of concentration on energy as we increase the electrolyte concentration. In the above figure, the concentration increases from curve 1 to 5.
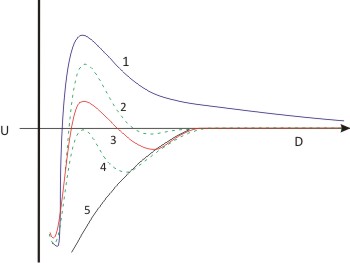
Fig. 10.4: Variation of Energy with solute concentration. point P represents CCC
|