These two terms in the total free-energy have quite different dependence on separation, D, which varies with different electrolyte concentrations. The Van-der waal Attraction term has inverse-square dependence whereas the Electrostatic Repulsion term has exponential decay.
A typical variation of energy with separation is depicted in the figure below-
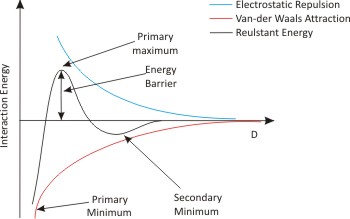
Fig. 10.3: Typical Energy barrier for two charged plates in an electrolytic medium
- The primary minimum indicates that the aggregated state is of the lowest-energy condition and this is where we would expect the particles to reside .
- The primary maximum acts as an activation barrier that must be exceeded for aggregation to occur. As two particles come closer, they must collide with sufficient energy to overcome the barrier provided by primary maximum.
- The secondary minimum could be seen as a flocculated state but the particles still have to cross the energy barrier to come into close contact at the minimum energy state.
To pose a suitable barrier to aggregation, the primary maximum must be at least 10kBT in order to achieve a stability which can be relied upon over an extended period of time.
|