Nucleation in liquid
Crystallization of liquid to solid is initiated by cooling of the melt.
In this case, from the Clausius-Clapeyron relation we can write,
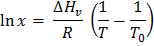
where 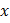 is defined as, is defined as, 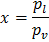 ; ratio of liquid to solid equilibrium vapor pressure. ; ratio of liquid to solid equilibrium vapor pressure.
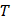 and and 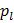 are a corresponding temperature (in units of kelvins or other absolute temperature units) and equilibrium vapor pressure of the liquid are a corresponding temperature (in units of kelvins or other absolute temperature units) and equilibrium vapor pressure of the liquid
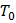 and and 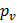 are the corresponding temperature and pressure of the solid at another point are the corresponding temperature and pressure of the solid at another point
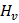 is the molar enthalpy of vaporization is the molar enthalpy of vaporization
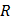 is the universal gas constant is the universal gas constant 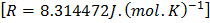
This is valid for 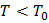 , which indicates that subcooling is needed. , which indicates that subcooling is needed.
At equilibrium we can write,
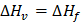 (heat of fusion) or, molar enthalpy of vaporization. (heat of fusion) or, molar enthalpy of vaporization.
Then we can get,
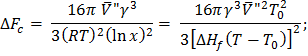
where 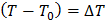 represents the degree of sub cooling, represents the degree of sub cooling, 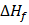 is heat of fusion. is heat of fusion.
|