39.2 POLYVINYL CHLORIDE (PVC)
Polymer Chemistry : The manufacture of Polyvinyl Chloride (PVC) follows addition type kinetics and produces linear polymers. Its molecular formula is given as –
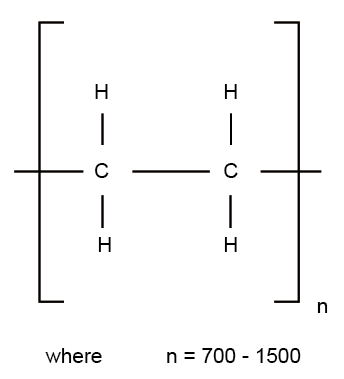
The vinyl chloride monomer (VCM) has a boiling point of 13.4°C and is a gas at room temperature and pressure. The vapor pressure of VCM over the typical polymerization temperature range of 50°C to 70°C is 800 – 1250 KPa.
Technology (Figure 39.2):
The two most commercially use methods for the manufacture of PVC are Emulsion Polymerization and Suspension Polymerization. The Suspension Polymerization process provides 80 % of the world production.
The Suspension Polymerization process is actually a bulk polymerization process which is carried out in millions of droplets . Each of these droplets act as small reactors. The liquid vinyl chloride is dispersed in water by vigorous stirring in a reactor. The reactor is fitted with baffles for optimum agitation and also has a condenser for heat removal. In the reactor , small droplets of size 30 - 40 μm diameter are formed.
A monomer soluble free radical initoiator is charge dinto the reactors. After charging , the reactor temperature is increased to 45 – 75°C . The heat decomposes some of the initiators to free radicals , and the monomers in these droplets begin to polymerize., the reaction is highly exothermic and the heat is removed via cooling jackets or by boil – off to the condenser. Thereafter, the condensed monomer is returned to the reactor. Although the PVC is insoluble in its monomer, it is swollen by VCM to form a coherent gel. Even in the gel phase , the polymerization continues. The polymerization is rapid at first, but slowly, as the conversion reaches 80 – 85 %, the rate is reduced due to monomer starvation.
At a predetermined pressure , the reaction is ended by adding a chain terminator or by venting the unreacted monomer to a recovery plant. Even after venting, the aqueous slurry contains 2 – 3 % unreacted monomer, which is then removed by stripping in a Stripping Column. The unreacted monomer is recovered and stored for later polymerization stages. The slurry is then passed through a heat exchanger and is passed through a continuous centrifuge to give a wet cake with 20 – 30 % moisture.
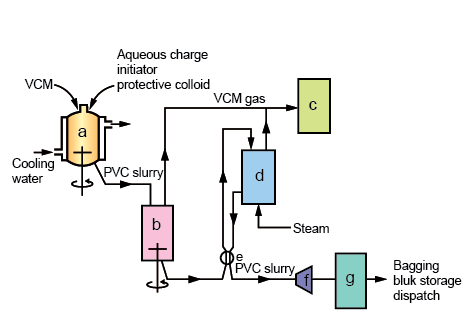
Figure 39.2. Manufacture of Poly(Vinyl Chloride)
a)Reactor; b)Blowdown Vessel; c)VCM Recovery Plant; d)Stripping Column; e)Heat Exchanger; f)Centrifuge; g)Driers