28.1 Introduction
Sucrose is a disaccharide that occurs naturally in most fruits and vegetables.
Sugar occurs in greatest quantities in sugarcane and sugar beets from which sugar is separated economically and commercially.
Chemical formula – c12H22O11
Molecular weight – 342
Density = 1.58 kg/m3
Sucrose is soluble in water but slightly soluble in methyl alcohol and ethyl alcohol.
Process flow sheet: Illustrated in Figure.
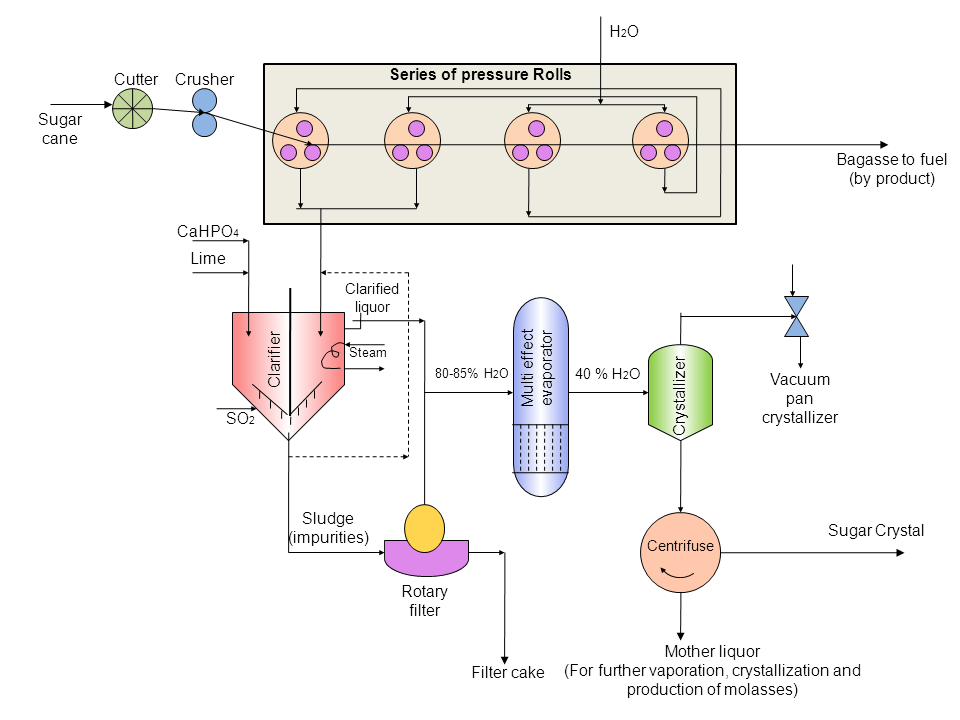
Figure 28.1 Flow sheet of manufacture of sugar from sugarcane
Raw material: Sugar cane
28.2 Functional role of various processes (Figure 28.1)
(a) Cutter
- • The cutter consists of knives on a cylindrical shaft which rotate at a velocity of 400 to 500 rpm.
• The knives cut the canes into small pieces.
(b) Crusher
- • Canes are shredded here.
• It consists of two rollers rotating in opposite direction.
(c) Series of Pressure mills
- • Crushed canes are passed through four pressure mills to extract juice.
• Each pressure mill is made up of cast iron rolls.
• Rolls are grooved and the width decreases from first roll to the last.
• Make up water added in the third and fourth mill is recycled back to the first two mills.
• About 85-90% of juice present in cane is extracted.
• Bagasses are produced as byproduct.
- • In general two methods of clarification are available for the manufacture of white sugar, namely, Sulfitation process and Carbonation process.
• The juice now comes to thickener.
• To precipitate the colloids, calcium phosphate(CaHPO4) is added followed by milk of lime.
• The milk of lime used has 9 to 10% strength and about 400mg CaO/litre alkalinity.
• Apart from maintaining pH about 7, SO2 gas also acts as a bleaching agent.
• Phosphoric acid or CO2 can also be substituted as acidifying agent depending upon the type of extracted juice.
• At the bottom of clarifier, mud (impurities) are settled and drained.
• Steam is used to slightly heat the juice.