3. Microkinetic modeling
Microkinetic modeling is used to link molecular-level informations about reactants, products and reactive intermediates on heterogeneous surfaces obtained by atomistic simulations and electronic structure calculations to macroscopic physical and chemical phenomena in systems involving chemical transformations. It is done using model parameters such as reaction rates, reactant conversion, product yields and selectivities predicted by the previous levels.
In this method no rate-determining mechanistic step (RDS) is assumed in contrast to more traditional models such as power law model or Langmuir– Hinshelwood–Hougen–Watson model. The mechanism for the reaction system is predicted based on experimental and theoretical study. All probable elementary steps are included. The rate constant for each of the elementary steps are specified from experiment or theory and the mechanism is then combined with the appropriate reactor design equations. The equations are solved to obtain relative reaction rates, coverage of surface intermediates, reactant conversion, product yields and selectivities. This is much more realistic method than traditional models where assumption of RDS is required. Non requirement of initial assumption of RDS in this method is more accurate as the RDS can change with reaction conditions. However accurate parameters for all forward and reverse reactions are needed to solve the equations of the model. Thus result in requirement of huge amount of informations about interactions of chemical species with catalysts. This is one of the major disadvantages of microkinetic modeling and resulted in limitation of its usage. At present with simultaneous advances in spectroscopic, isotopic tracing and other experimental methods, obtaining detailed informations has become more feasible and consequently quantum chemical techniques and microkinetic models are being used more frequently.
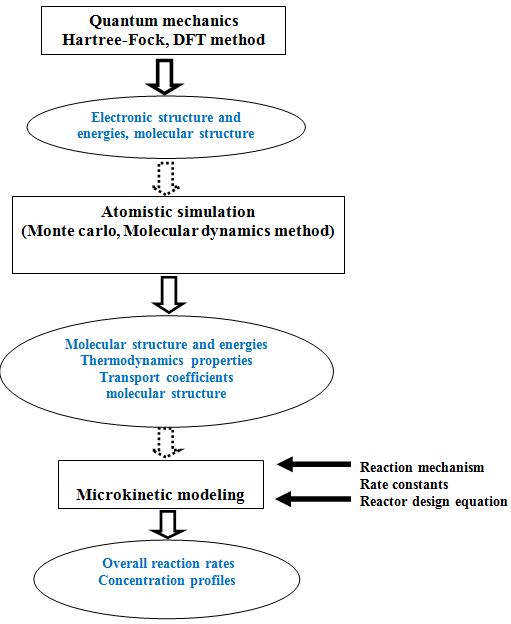
Fig 1. Flow diagram showing different levels of catalytic modeling
Reference:
• Linda J. Broadbelt, Randall Q. Snurr, Applications of molecular modeling in heterogeneous catalysis research, Applied Catalysis A: General 200 (2000) 23–46
• H. Höltje, W. Sippl, D. Rognan, G. Folkers, Molecular Modeling: Basic Principles and Applications, Wiley-VCH, 2008
Journal reference
1. C. A.Wegermann, J. C. Rocha, S. M. Drechsel, F.S. Nunes, Dyes and Pigments 99(2013)839-849
2. X. Ma, H. H. Schobert, J. of Molecular Catalysis A: Chemical 160(2000)409–427
3. J. L.C. Fajin, M. Natalia, D.S. Cordeiro, J. R.B. Gomes , Applied Catalysis A: General 458 (2013) 90– 102
4. M. D'Amore, R. Credendino, P.H.M. Budzelaar, M. Causa, V. Busico, J.of Catalysis 286 (2012) 103–110