| |
Assignment of Chapter 4
(Answers are in parenthesis) |
-
A rigid vessel of 0.06 m3 volume contains an ideal gas, CV = (5/2)R, at 500 K and 1 bar. (a) If 15 kJ of heat is transferred to the gas, determine its entropy change. (b) If the vessel is fitted with a stirrer that is rotated by a shaft so that work in the amount of 15 kJ is done on the gas, what is the entropy change of the gas if the process is adiabatic? What is ΔStotal ? (20.8J/k, 20.8J/K)
-
An ideal gas, Cp= (7/2)R, is heated in a steady-flow heat exchanger from 70°C to 190°C by another stream of the same ideal gas which enters at 320°C. The flow rates of the two streams are the same, and heat losses from the exchanger are negligible. Calculate the molar entropy changes of the two gas streams for both parallel and countercurrent flow in the exchanger. What is ΔStotal in each case? (2.15J/molK, same in both cases)
-
One mole of an ideal gas, Cp = (7/2)R and CV = (5/2)R, is compressed adiabatically in a piston cylinder device from 2 bar and 25°C to 7 bar. The process is irreversible and requires 35% more work than a reversible, adiabatic compression from the same initial state to the same final pressure. What is the entropy change of the gas? (2.914J/molK)
-
A mass m of liquid water at temperature T1 is mixed adiabatically and isobarically with an equal mass of liquid water at temperature T2. Assuming constant Cp, show that: 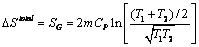 and prove that this is positive. What would be the result if the masses of the water were different, say, m1 and m2? and prove that this is positive. What would be the result if the masses of the water were different, say, m1 and m2?
-
A reversible cycle executed by 1 mol of an ideal gas for which Cp = (5/2)R and CV = (3/2)R consists of the following: Starting at T1 = 700 K and P1 = 1.5 bar, the gas is cooled at constant pressure to T2 = 350 K. From 350 K and 1.5 bar, the gas is compressed isothermally to pressure P2. The gas returns to its initial state along a path for which PT = constant. What is the thermal efficiency of the cycle? (0.07)
-
One mole of an ideal gas is compressed isothermally but irreversibly at 130°C from 2.5 bar to 6.5 bar in a piston cylinder device. The work required is 30% greater than the work of reversible, isothermal compression. The heat transferred from the gas during compression flows to a heat reservoir at 25°C. Calculate the entropy changes of the gas, the heat reservoir, and ΔStotal. (–7.94J/molK, 13.96J/molK, 6.02J/molK)
-
Ten kmol per hour of air is throttled from upstream conditions of 25°C and 10 bar to a downstream pressure of 1.2 bar. Assume air to be an ideal gas with Cp= (7/2)R. (a) What is the downstream temperature? (b) What is the entropy change of the air in J/molK? (c) What is the rate of entropy generation in W/K? (d) If the surroundings are at 20°C, what is the lost work? (298K, 17.63J/molK, 48.9W/K, 5.2kJ/mol)
-
A steady-flow adiabatic turbine (expander) accepts gas at conditions T1 = 500 K, P1 = 6 bar, and discharges at conditions T2 = 371 K, P2 = 1.2 bar. Assuming ideal gases, determine (per mole of gas) Wactual, Wideal, Wlost, and entropy generation rate.Tsurrounding, = 300 K, Cp/R = 7/2. (3753.8J, –5163J, 1409J, 4.7J/K)
-
An ideal gas at 2500 kPa is throttled adiabatically to 150 kPa at the rate of 20 mol/s. Determine rates of entropy generation and lost work if Tsurrounding = 300 K (0.468kW/K, 140.3kW)
-
A vessel, divided into two parts by a partition, contains 4 mol of nitrogen gas at 75°C and 30 bar on one side and 2.5 mol of argon gas at 130°C and 20 bar on the other. If the partition is removed and the gases mix adiabatically and completely, what is the change in entropy? Assume nitrogen to be an ideal gas with Cv = (5/2)R and argon to be an ideal gas with Cv = (3/2)R. [38.3J/K]
-
A stream of nitrogen flowing at the rate of 2 kg s-1 and a stream of hydrogen flowing at the rate of 0.5 kgs-1 mix adiabatically in a steady-flow process. If the gases are assumed ideal, what is the rate of entropy increase as a result of the process? [1411W/K]
|
 Back Back |
|
|
|