Thus we see that the results of both eqns. A.4.1.2 and A.4.1.10 are identical. There is thus a convergence between the macroscopic and microscopic description of the concept of entropy.
It is particularly significant that the above result obtains if one assumes the highest value of 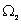 as its most likely value. It may be argued that the highest value of as its most likely value. It may be argued that the highest value of 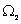 corresponds to the most disordered state at the molecular level. Indeed extending the argument, since corresponds to the most disordered state at the molecular level. Indeed extending the argument, since 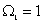 the initial state is more ordered than state 2, as in the former all molecules are characterized by a single position coordinate, and therefore the probability of finding a molecule in that position is unity; in other words one has complete (or ‘certain’) information about position of any molecule. In contrast in the second state the probability of finding a molecule at any one position is half. This obtains if each compartment contains exactly half the total number of molecules. Any other distribution would be less disordered than this state, and also less probable! This is the reasoning which lies at the root of the popular description of entropy as a measure of disorder at the molecular level. It follows that a system at non-equilibrium state (say, at the point of rupture of the partition) always tends to the most disordered molecular state when it attains a new equilibrium. Since one has less microscopic information about a system with a greater degree of disorder, ‘information’ is also described as negentropy. the initial state is more ordered than state 2, as in the former all molecules are characterized by a single position coordinate, and therefore the probability of finding a molecule in that position is unity; in other words one has complete (or ‘certain’) information about position of any molecule. In contrast in the second state the probability of finding a molecule at any one position is half. This obtains if each compartment contains exactly half the total number of molecules. Any other distribution would be less disordered than this state, and also less probable! This is the reasoning which lies at the root of the popular description of entropy as a measure of disorder at the molecular level. It follows that a system at non-equilibrium state (say, at the point of rupture of the partition) always tends to the most disordered molecular state when it attains a new equilibrium. Since one has less microscopic information about a system with a greater degree of disorder, ‘information’ is also described as negentropy. |