Addition of low amount of salt to the protein solution results in the displacement of bonded water molecule with an increase in protein solubility (Figure 31.2). This effect is called as “salting-in”. In the presence of more amount of salt, water molecule shielding protein side chains are displaced completely with an exposure of hydrophobic patches on protein surface to induce protein precipitation or decrease in protein solubility. This effect is called as “salting-out”. The phenomenon of salting out is modulated so that addition of salt induces exposure of hydrophobic patches on protein but does not cause precipitation or aggregation. The exposure of hydrophobic patches facilitates the binding of protein to the non-polar ligand attached to the matrix. When the concentration of salt is decreased, the exposed hydrophobic patches on protein reduces the affinity towards matrix and as a result it get eluted (Figure 31.3).
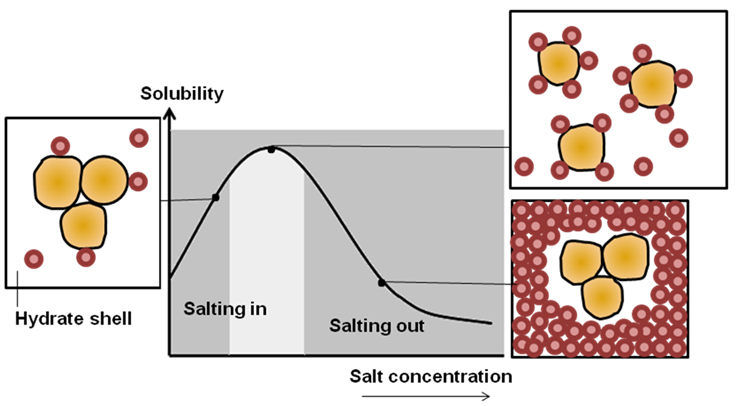
Figure 31.2: Effect of salt on protein, salting in and salting out effect.
The choice of HIC gel- The different commercially available HIC matrix are given in Table 31.1. Choosing a suitable HIC matrix is essential to achieve best result. The strength of the binding of analyte on a HIC column is governed by the length of the aliphatic linear ligand. Matrix with aromatic ring containing ligand makes additional Π-Π interaction and they will bind analyte more strongly than same number of carbon aliphatic ligand. In addition, presence of Π-Π interaction gives selectivity as well, such as ring containing aromatic ligand, phenylalanine. At last, ligand density plays a vital role in the strength of binding of an analyte to the matrix. Hence, these points should be consider to choose a suitable matrix for purification.