(c) Electron ionization
Electron Ionization (EI) works well for many gas phase molecules, but it results in extensive fragmentation and molecular ions are not observed for many compounds. Fragmentation mass spectra are sometime useful because it provides structural information of a molecule.
The electron beam is produced by a filament of rhenium or tungsten wire by thermionic emission. When cathode filament of rhenium or tungsten is heated at temperature over 1000 K, electrons are emitted. The generated electrons are accelerated to 70 eV which results in electron beam. The volatile sample or sample in gaseous phase containing neutral molecules is introduced to the ion source in a perpendicular direction to the electron beam. Electron impact on the analyte results in either loss of electron (to produce cation) or gain of electron (to produce anion). Chemical bonds in organic molecules are formed by pairing of electrons. Electron impact may knock out one of the electron. This leaves the bond with a single unpaired electron. This is radical as well as being cation written as M+., where (+) indicates ionic state while ( . ) indicates radical. Electron impact may result in electron capture (extra unpaired electron). This generates a radical as well as being anion written as M+., where (-) indicates ionic state while ( . ) indicates radical (Fig. 5.)
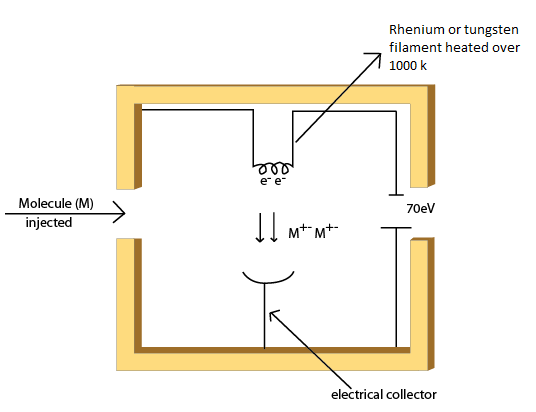
Figure 5: A schematic of the mechanism of ion formation in electron ionization
|