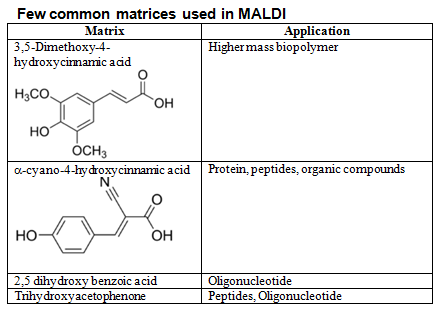
Table 1: Few common matrices used in MALDI
(b) Electrospray Ionisation (ESI)
Electrospray Ionisation (ESI) is a preferred method of ionization when the sample is in liquid form. This is also a soft method of ionization and results in less fragmentation. ESI is a very valuable method for analysis of biological samples. The method was developed by John Fenn and he shared 2002 Nobel prize in chemistry for this work. The analyte is introduced either from a syringe pump or as the eluent flow from liquid chromatography with a flow rate 1μl min -1 . The analyte solution passes through the electrospray needle ( Stainless steel capillary with 75-150 1μm internal diameters) that has a high potential difference (with respect to the counter electrode) applied to it (typically in the range from 2.5 to 4 kV). This forces the spraying of charged droplets from the needle with a surface charge of the same polarity to the charge on the needle. As droplet moves towards counter electrode cone (which passes it to analyzer), solvent evaporation occurs and droplet shrinks until it reaches the point that the surface tension can no longer sustain the charge (the Rayleigh limit) and at that point droplets break. This produces smaller droplets and the process is repeated. Finally after all solvent evaporated, charge is passed on to analyte. These charged analyte molecules can have single or multiple charges (Fig. 4).

Figure 4: A schematic of the mechanism of ion formation in ESI.
|