1. So, at the thermal equilibrium, the pressure 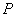 and volume and volume 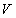 are inversely proportional to each other, i.e. are inversely proportional to each other, i.e.
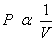 --- Boyle's Law. --- Boyle's Law.
2. Similarly, by simple experiments we can observe that at constant pressure, volume of the gas is directly
proportional to the temperature and at constant volume, pressure of the gas is directly proportional to the
temperature.
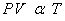 --- Charles Law. --- Charles Law.
We can summarize the above two observations to the following relations,
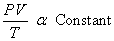 ................................... ( 2 ) ................................... ( 2 )
If we take two different kinds of gases at same pressure, volume and temperature, then the quantity of the gas should be the same. The quantity of the gases can be better represented by the number of molecules. So, it is clear that, the constant in equn(2) is related to the number of molecules. So, equn(2) can be written as
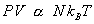 ................................... ( 3 ) ................................... ( 3 )
where 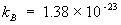 joule/K is Boltzmann constant. joule/K is Boltzmann constant. |