| |
Calculate the energy released in the fission reaction |
|
The masses of the reactants and the product (in units of u)are as follows : |
| |
mass of 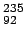 U U |
= |
235.043929 |
mass of 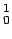 n n |
= |
1.008665 |
|
|
------- |
| Total mass of reactants |
= |
236.052594 |
|
| |
mass of 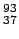 Rb Rb |
= |
92.922042 |
mass of 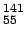 Cs Cs |
= |
140.920046 |
mass of 2 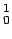 n n |
= |
2.017330 |
|
|
------- |
| Total mass of product |
= |
235.859418 |
|
| |
The products being lighter than the reactants by 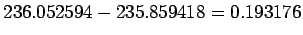 u. This amounts to u. This amounts to  MeV of energy. MeV of energy. |
| |
Fission produces about 200 Mev of energy of which about 175 MeV is the kinetic energy of fission fragments, the reamaining energy is distributed as the kinetic energy of neutrons and energy associated with photons, neutrinos and other radioactive products. |