 |
The Helium nucleus 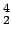 He (also called the alpha particle ) has a mass of 4.001 506 u. Determine its binding energy in terms of the atomic mass units and in MeV. He (also called the alpha particle ) has a mass of 4.001 506 u. Determine its binding energy in terms of the atomic mass units and in MeV. |
| |
Alpha particle has two protons and two neutrons. Binding energy calculation in atomic mass unit is shown alongside. In terms of MeV, the binding energy is  MeV, i.e. 7.074 MeV per nucleon. |