 |
If an electron makes a transition from 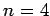 to to 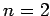 , determine (i) the wavelength of emitted radiation and (ii) the recoil speed of the electron. , determine (i) the wavelength of emitted radiation and (ii) the recoil speed of the electron. |
|
The wavelength of emitted radiation is
|
| |
The wavelength is 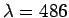 nm. The momentum associated with this radiation is nm. The momentum associated with this radiation is |
| |
By conservation of momentum, this is also the magnitude of the momentum imparted to the atom as a whole. The recoil speed of the electron is |