 |
A monochromatic source of light with a wavelength 200 nm and power output of 2 watts is held at a distance of 0.1m from the surface of an aluminium foil. Aluminium has a work function of 4.2 eV and an atomic radius of 0.15 nm. Take the photo-emission efficiency to be 2.5%. Calculate |
|
the kinetic energy of the fastest and the slowest photoelectron emitted, |
|
the average number of photons falling on an atom of Al per second, |
|
the number of photoelectrons emitted per unit area per second. |
|
200 nm corresponds to a photon energy of 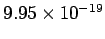 J, which is equal to 6.2 eV. Thus the kinetic energy J, which is equal to 6.2 eV. Thus the kinetic energy |
| |
of the fastest electron is 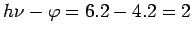 eV. The kinetic energy of the slowest electron is zero. eV. The kinetic energy of the slowest electron is zero. |
|
The light falling on an unit area at a distance 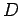 from the source has an intensity of from the source has an intensity of |
| |
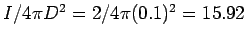 W/m W/m 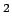 . The amount of radiation captured by an atom of radius . The amount of radiation captured by an atom of radius 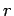 is is |
| |
The number of photons is obtained by dividing this by the energy of a single photon. Thus the number of photons captured by an atom on the foil is 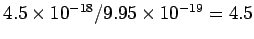 per second. per second. |
|
The photo-emission efficiency is the ratio of the number of photoelectrons emitted from a surface to the number |
| |
of photons falling on the surface in a given time. The number of electrons falling on unit area of the foil is 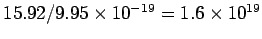 per second. With 2.5% efficiency, the number of electrons emitted is per second. With 2.5% efficiency, the number of electrons emitted is 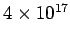 /m /m  -s. -s. |