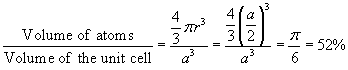Example 1: Calculate the maximum fraction of the volume in a simple cubic crystal occupied by the atoms. Assume that the atoms are closely packed and that they can be treated as hard spheres. This fraction is also called the packing density.The atoms in a simple cubic crystal are located at the corners of the units cell, a cube with side a. Adjacent atoms touch each other so that the radius of each atom equals
. There are eight atoms occupying the corners of the cube, but only one eighth of each is within the unit cell so that the number of atoms equals one per unit cell. The packing density is then obtained as follows: