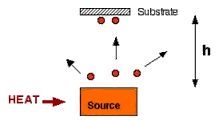
Fig. 3.22: Conversion of source material to gaseous form
In the first stage, as shown in Fig. 3.22 the gaseous state is achieved by heating the source so that its vapor pressure (Pvapor) is greater than 10-4 torr. The evaporation is carried out in standard base vacuum pressures and the evaporation rate (flux) can be estimated from kinetic theory described in previous section. Some sources sublime from solid while others evaporate from liquid. When source compounds are changed to vapor, it may break apart and produce films with different stoichiometry. For example instead of SiO2, one may end up with SiOx, where x < 2. Similarly, metal alloy sources do not give same alloy in film; its components evaporate independently based on each separate vapor pressure and the composition of alloy source also changes with time.