The number of gas molecules colliding with a surface each second is given by
= 0.25 n vrms , where
is the collision rate of gas molecules, n is the number of molecules per unit volume and vrms is the average velocity of a gas molecule. Expressing this equation in terms of things one can directly measure:
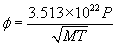
Where
is in molecules/cm2 –sec, P is the pressure in torr, M is the molecular weight of the gas molecule and T is the temperature in K. As an example, Nitrogen (N2) has a molecular weight M = 28. In a chamber with nitrogen at room temperature (293 K) and a pressure of 1 x 10-7 torr,
= 3.88 x 1013 molecules/cm2-sec.