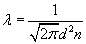Actually, we need to consider that all the molecules are moving. Clearly, what really counts in the collision rate is the relative velocity of the molecules. Defining the average velocity as the root mean square velocity, if one molecule has velocity v1 and another has v2, then the square of the relative velocity, since
must average to zero, the relative directions being random. So the average square of the relative velocity is twice the average square of the velocity, and therefore the average root-mean-square velocity is up by a factor v2, and the collision rate is increased by this factor. Consequently, the mean free path is decreased by a factor of
.