In the absence of an external battery (or illumination), the continuous supply of holes to the reaction interface ceases. The concentration of the thermally generated holes is insufficient for the formation of SiO2 beyond a few monolayers. However, the battery provides the necessary holes for this anode reaction that sustains the process of oxide growth. The H+ drifts to the cathode, where it is evolved as molecular hydrogen by the addition of electrons
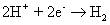
The anodic oxidation of p-type material is relatively straightforward since there is no problem with delivering holes to the semiconductor surface by means of a battery. The situation is somewhat complex for n-type semiconductor. Here the initial charge transfer from the semiconductor into the electrolyte creates a depletion layer in semiconductor, and a barrier to the flow of holes. In effect, then, the electrolytic-semiconductor system behaves much like a Schottky diode with the electrolyte serving the role of the metal. In order to sustain the process of oxide growth, provision is to be made to supply holes to the semiconductor surface. One approach is to illuminate the sample to provide these holes by photo-generation. Alternately, the anodisation cell can be operated at a voltage, which exceeds the Schottky diode breakdown voltage, so that avalanche generated holes can allow the oxidation to proceed.