- Conductivity in between those of metals and insulators.
- Conductivity can be varied over orders of magnitude by changes in
temperature, optical excitation, and impurity content (doping).
- Generally found in column IV and neighboring columns of the periodic
table.
- Elemental semiconductors: Si, Ge.
- Compound semiconductors:
Binary :GaAs, AlAs, GaP, etc. (III-V). ZnS, ZnTe, CdSe (II-VI). SiC, SiGe (IV compounds).
Ternary : GaAsP.
Quaternary : InGaAsP.
- Si widely used for rectifiers, transistors, and ICs.
- III-V compounds widely used in optoelectronic and high-speed applications.
- Integrated circuits (ICs) SSI, MSI, LSI, and VLSI.
- Fluorescent materials used in TV screens II-VI (ZnS).
- Light detectors InSb, CdSe, PbTe, HgCdTe.
- Infrared and nuclear radiation detectors Si and Ge.
- Gunn diode (microwave device) GaAs, InP.
- Semiconductor LEDs GaAs, GaP.
- Semiconductor LASERs GaAs, AlGaAs.
- Distinguishing feature among metals, insulators, and semiconductors.
- Determines the absorption/emission spectra, the leakage current, and
the intrinsic conductivity.
- Unique value for each semiconductor (e.g. 1.12 eV for Si, 1.42 eV for GaAs) function of temperature.
- Can be added in precisely controlled amounts.
- Can change the electronic and optical properties.
- Used to vary conductivity over wide ranges.
- Can even change conduction process from conduction by negative charge
carriers to positive charge carriers and vice versa.
- Controlled addition of impurities doping.
- Semiconductor properties can be strongly affected by crystal structure.
- Basically, there are three types of solids: crystalline, amorphous, and polycrystalline.
- Atoms making up the solid arranged in a periodic fashion, repeated
throughout.
- Have long-range order.
- Used for IC fabrication.
- Have no periodic structure at all.
- Interatomic distance and bond angles are almost the same as in the
crystalline material of the same substance, however, a long-range order
is missing.
- Said to have short-range order.
- a-Si (alloy of amorphous Si with
 and other similar amorphous alloys) has found important applications
in photovoltaic technology and in large-area ICs used in flat displays,
printers, copiers, scanners, and imagers.
and other similar amorphous alloys) has found important applications
in photovoltaic technology and in large-area ICs used in flat displays,
printers, copiers, scanners, and imagers.
- Composed of many small regions of single-crystal material of irregular size, separated by grain boundaries.
- 3-D periodic arrangement of atoms in a crystal.
- Defined by primitive basis vectors
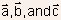 ,
which are three independent shortest vectors connecting lattice sites.
,
which are three independent shortest vectors connecting lattice sites.
- The coordinates of all points belonging to the crystal lattice are
given by vectors
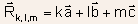 , where k, l, and m are integers.
, where k, l, and m are integers.
- Properties of the periodic crystal determine the allowed energies of electrons that participate in the conduction process. Thus, the lattice not only determines the mechanical properties of the crystal, but also its electrical properties.
- Representative of the entire crystal and regularly repeated throughout
the crystal.
- The crystal can be analyzed as a whole by investigating a representative
volume.
- Can find:
i) the distances between nearest atoms and next nearest atoms,
ii) the fraction of the unit cell volume filled by atoms, and
iii) the density of the solid (related to the atomic arrangement).
- Smallest unit cell that can be repeated to form the lattice.
- Simplest 3-D lattice, where the unit cell is a cube.
- Three types:
i) simple cubic (sc) (e.g., Ga),
ii) body-centered cubic (bcc) (e.g., Na, W), and
iii) face-centered cubic (fcc) (e.g., Al, Au).
- Lattice constant: the length of each side of the cube.
- Two interpenetrating fcc sublattices spaced 1/4th along the body diagonal.
- When the constituent atoms of the two sublattices are different, then the structure is Zincblende (e.g. GaAs).
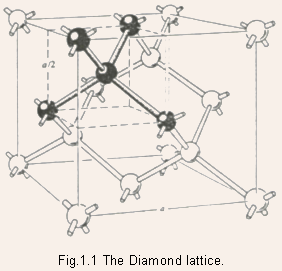
- Diamond and Zincblende are the two most common crystal structures
for cubic semiconductors.
- Each atom in diamond and zincblende lattice is surrounded by four
nearest neighbors.
- Tetrahedral configuration.
- By varying the atomic compositions of these two sublattices, one can
grow ternary (e.g.
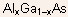 )
and quaternary (e.g.,
)
and quaternary (e.g., 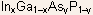 )
compounds.
)
compounds.
EXAMPLE 1.1: Find the fraction of the unit cell volume filled with hard spheres for a diamond lattice.
SOLUTION: Diamond lattice unit cell consists of an fcc lattice along with 4 inside atoms. The corner atoms in an fcc lattice is shared by seven nearest neighbors, thus each contributing 1/8th of a sphere, and the face atoms are shared by one nearest neighbor, thus each contribution ½ of a sphere.
Thus, Atoms/cell = 1 (corners) + 3 (faces) + 4 (inside) = 8 Nearest neighbor distance = (
Maximum fraction of cell filled =
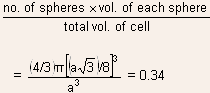
Therefore, the unit cell volume filled with hard spheres for a diamond lattice is only 34%, thus, the diamond lattice is relatively loosely packed.
Planes and Directions
- Found by taking the reciprocal of the intercepts of the plane with
the coordinate axes, and converting these to integers.
- Define a set of parallel planes.
- (hkl) a particular plane.
- {hkl} equivalent (hkl) planes.
- [hkl] a particular direction, perpendicular to the (hkl) plane
for cubic crystals.
- <hkl> equivalent [hkl] directions.
- (hkl) a particular plane.
- Negative intercepts/directions are denoted by placing bars above the
integers, e.g.
 .
.
- Three main indices (100), (110), and (111).
- Atom densities different for different planes different electronic properties.
EXAMPLE 1.2: Show a (643) plane and (643) direction in a cubic lattice.
SOLUTION: (643) plane: reciprocals (1/6,1/4,1/3), converting them to smallest set of integers gives (2,3,4); thus, the plane has (x,y,z) intercepts as (2,3,4).
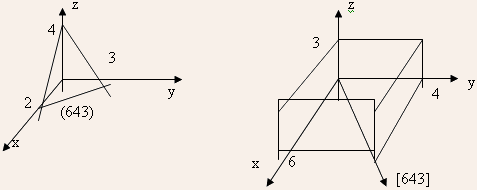
1.1 Determine the nearest neighbor distance, and the number of the nearest and the next neighbors of sc, bcc, fcc, and diamond lattices with lattice constant a.
1.2 Determine the maximum fractions of the unit cell volume that can be filled by hard spheres in the sc, bcc, and fcc lattices.
1.3 Clearly state the equivalent {100}, {110}, and {111} planes and determine their total numbers in a cubic crystal.
1.4 Show the (211) plane and the [211] direction in a cubic crystal lattice.
1.5 Calculate the densities of Ge (diamond structure, a = 5.66
1.6 Determine the atom density (in number of atoms/cm2) in (100), (110), and (111) planes of Si (a = 5.43