| Fundamental concepts of semiconductors |
| |
| |
| |
| |
| 1.4 Band gaps in semiconductors |
| |
Until now, we saw how electrons behave in a crystal and visualized, at least to
some extent, the energy band diagrams. However, a complete energy band diagram of a semiconductor is much more
complex than what we have seen. For example Si band diagram. Can you identify those symmetry point indications?
( go back to our previous discussion on symmetry point notations). Principally, the indexing follows the rules of
group theory displaying the symmetries (but we are not going to describe those details here).
In this section, we first take a closer look at the energy band diagrams of common semiconductors. We will also
see how the energy band gap varies with temperature. If you look at the Si band diagram shown in fig 2.17, the maximum
|
| |
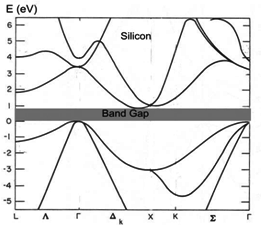
Fig. 2.17. Energy band diagram of silicon.
|
| Semiconductor |
E g (at 300K) in eV |
| Silicon (Si) |
1.11 |
| Germanium (Ge) |
0.66 |
| Gallium Arsenide (GaAs) |
1.41 |
| Indium phosphate (InP) |
1.34 |
| Zinc tellurite (ZnTe) |
2.26 |
| Cadmium Tellurite (CdTe) |
1.43 |
|
of the valence band (at Γ) does not coincide with the minimum of the conduction band .
In other words, the valence band has a minimum at a value of k (k1) , which is different from that of the k value (k2)
for the conduction band minimum.
|
|
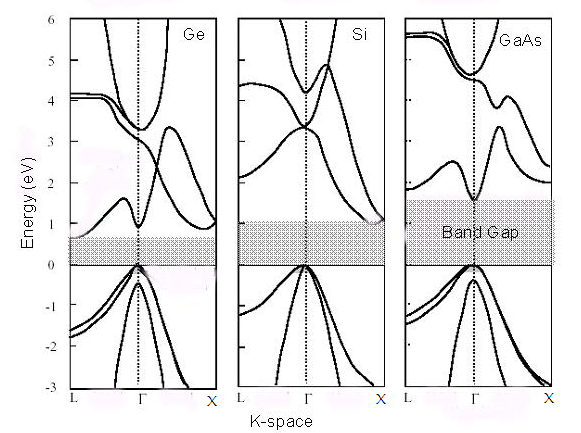
Fig2.18. Energy band diagram of silicon (Si), Germanium (Ge) and Gallium Arsenide (GaAs).
|
For an electron to excite from the valence band to the conduction band,
we need a change in momentum ( Δk = k1-k2). This is the so-called indirect band gap. This makes such semiconductors
more unlikely for a source of light, because the requirement of both the energy and momentum in optical process
leads to photon involvement. Similar is the case for Germanium. Whereas if we observe (Fig 2.18) the band structure
of GaAs, both maximum of valance and minimum of conduction band are on the same k vector, that is Γ. That means
there is no requirement of momentum change to produce a transition between valance band to the conduction band.
Such materials are called direct band gap materials. These direct band gap materials are efficient sources of light,
such as LEDs (Find semiconductors and their band gaps of those LED materials covering a wide spectrum
from 300nm-1500nm wavelength.). See the table for Eg values of some of the semiconductors used in the device technology.
|
| |
| |
| |
| |
| |
| |
| |

