| |
| |
Introduction to Quantum ideas |
|
| |
where 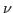 is the frequency of the incident radiation and is the frequency of the incident radiation and 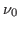 is the critical frequency below which no electrons are emitted. is the critical frequency below which no electrons are emitted.
(3) The electrons are emitted almost instantaneously without any
noticeable time delay.
These observations are not consistent with wave description of
e.m. radiation. The results suggest |
|
| |
|
|
| |
|
(2.3) |
| |
where the observed proportionality constant 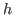 is Planck's constant.
Einstein's interpretation (1905) was that radiation comes in energy
quanta is Planck's constant.
Einstein's interpretation (1905) was that radiation comes in energy
quanta 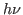 , photons, and their energy goes into overcoming the
minimum binding energy , photons, and their energy goes into overcoming the
minimum binding energy 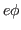 called the work function, and into the
kinetic energy of the emitted electrons. Some important points to be
noted are: called the work function, and into the
kinetic energy of the emitted electrons. Some important points to be
noted are:
(1) Ultraviolet radiation is needed since 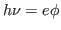 and and 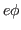 is
of the order of 3-5 eV for alkali metals. The value of is
of the order of 3-5 eV for alkali metals. The value of 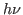 for
visible radiation is about 2.5 eV. for
visible radiation is about 2.5 eV.
(2) Alkali metal plates are needed since they have the lowest work
functions, 3-5 eV.
(3) Only a small fraction of incident radiation, 5 percent is
responsible for ejecting electrons, the rest is absorbed by the plate
as a whole.
(4) Photoelectric effect in atoms in the form of gases, is described
as photo-ionization. Here since there is no collective absorption,
number of photons absorbed is equal to the number of electrons
ejected.
(5)Energy for emission can come from heating, thermionic emission. In
this case the rate of emission is determined by statistical
properties in terms of 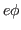 and temperature T. and temperature T.
(6) With the development of very high intensity radiation in the form
of lasers, now we can have multi-photon absorption for which one has
the relation |
| |
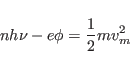 |
(2.4) |
| |
for energy conservation, where 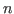 is the number of photons absorbed
by a single electron. is the number of photons absorbed
by a single electron. |
|
|
|
|