Now we will carry out some examples of diatomic molecules to find out the ground state from its electronic configuration.
Closed shell configuration:
For any closed shell molecular electronic configuration, for example 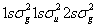 , there is no net orbital and spin angular momentum. So, L = 0 and S = 0. The term will be always, , there is no net orbital and spin angular momentum. So, L = 0 and S = 0. The term will be always, 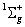 state. state.
One unpaired electron:
Let us take the case of hydrogen molecular ion 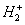
The electronic configuration 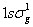 (figure-34.3). (figure-34.3). |