| |
A molecular term symbol labels the molecular states and specifies the total spin and orbital angular momentum of the molecule, along with various other symmetries.
The term symbol is written as
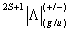
In the following we will understand the meaning of these notations. |
- As defined, L is the quantum number for the total orbital angular momentum L of the electrons about the inter-nuclear axis.
- As defined, S is the total spin angular momentum quantum number, formed from the individual electron. Spin quantum number of single electron s = 1/2 , and ms = ±1/2.
For one unpaired electron, S = s = 1/2 (a doublet state with 2S + 1 = 2).
For two unpaired electrons, the possible values are S = 1 and 0. For S = 1, gives triplet (2S + 1 = 3) and for S = 0 gives singlet (2S + 1 = 1) states.
- Symmetric molecules are having a centre of symmetry. The gerade (g) or ungerade (u) subscripts apply only to these molecules and labels the symmetry of the electronic wavefunction with respect to inversion through this centre. For the molecule, this can be thought of as simple multiplication of functions of even or odd symmetry of the individual electrons. In the following table provides the resultant symmetry of the molecule from two electrons.
|
Individual electrons |
Molecular |
gerade (g ) ´ gerade(g), even´even |
gerade, g |
Gerade (g) ´ ungerade (u), even´odd |
ungerade, u |
ungerade (u) ´ ungerade (u), odd´odd |
gerade, g |
|
- The +/− superscript applies only to å state (L = 0), and labels the symmetry of the electronic wavefunction with respect to reflection in a plane containing the nuclei.
|
|
|
|