Case-III
Let us first consider the case where 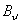 < <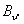
In this case, from equation-32.9
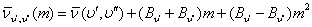
it can be seen that 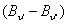 is negative quantity. So for m negative i.e P-branch, the separation will go on increasing. On the other hand, for m positive, the sign of the second and the third term will be opposite. Hence for R-branch the spacing between the lines will decrease and slowly the lines will merge. This forms the band head R-branch which is the vertex of the parabola as shown in figure-32.4. is negative quantity. So for m negative i.e P-branch, the separation will go on increasing. On the other hand, for m positive, the sign of the second and the third term will be opposite. Hence for R-branch the spacing between the lines will decrease and slowly the lines will merge. This forms the band head R-branch which is the vertex of the parabola as shown in figure-32.4. |