The other px, and py orbitals overlap side-by-side. This is known as p-bonding. This overlap is less than the overlap along the bond axis.
Note that, the overlap is more ® it will be more stable and thus energy will be lower.
Thus, the p-bonding orbital will be higher in energy than that of the s-orbital.
p-orbitals are asymmetric with respect to the bond axis as shown in figure-27.4. No electron density surrounding the bond axis. It has node along the internuclear axis.
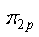 is asymmetric about the center of inversion, and thus is asymmetric about the center of inversion, and thus 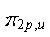
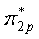 is symmetric about the center of inversion, and thus is symmetric about the center of inversion, and thus 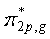 |