We have seen in the previous lecture that H2+-ion is stable because of the bonding between the two hydrogen atoms which form a bound state with one electron.
Similar to this, if we consider two hydrogen atoms to form hydrogen molecule, we expect that the wavefunctions of two hydrogen atoms 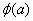 and and 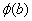 will interact constructively or destructively. will interact constructively or destructively.
If they interact constructively then the molecular wavefunction
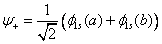
This will form bonding orbital. And if they interact destructively, it will form antibonding orbital
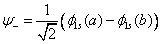
We have also realized that the degenerate levels of the two hydrogen atoms will be separated as the two nuclei come closer to each other.
As shown in figure 27.1, the ground state of two hydrogen atomic orbitals split into two energy levels – bonding 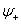 and anitibonding and anitibonding 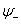 . . |