So, 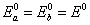
If we let the nuclei approach one another, the electron, which was, for example, at first attached to nucleus 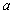 , will experience the attractive Coulomb force of nucleus , will experience the attractive Coulomb force of nucleus 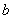 . .
Conversely, an electron which was at first bound to nucleus 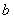 will experience the attractive Coulomb force of nucleus will experience the attractive Coulomb force of nucleus 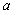 . We must therefore set up a Schrodinger Equation which contains the Coulomb potential for both. . We must therefore set up a Schrodinger Equation which contains the Coulomb potential for both.
Further in order to calculate the total energy of the system, we must take into account the Coulomb repulsion between nuclei. The additional energy 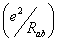 is not directly related to the energy of the electron, it will only produce a constant shift of all the energy eigen values. We will introduce it at the end. is not directly related to the energy of the electron, it will only produce a constant shift of all the energy eigen values. We will introduce it at the end. |