To compare the differences between the functions 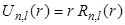 and and 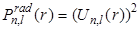 for the n = 1 and n = 2 states of hydrogen, they are plotted on the same graph as shown in the following Figure - 9.9 and Figure - 9.10: for the n = 1 and n = 2 states of hydrogen, they are plotted on the same graph as shown in the following Figure - 9.9 and Figure - 9.10:
- The increase in l by 1 from 2s to 2p states means that neither
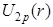 nor nor 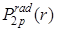 have nodes. Asymptotically, both functions decay at the same rate as their 2s counterparts, because the decay constant for a pure- Coulomb potential energy doesn't depend on l. have nodes. Asymptotically, both functions decay at the same rate as their 2s counterparts, because the decay constant for a pure- Coulomb potential energy doesn't depend on l.
- The sole peak in
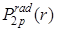 occurs at r = 4 occurs at r = 4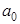 , where its value is 0.19. Interestingly, the mean radius of 2p state = 5 , where its value is 0.19. Interestingly, the mean radius of 2p state = 5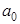 is 1 is 1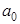 smaller than that of the 2s state. smaller than that of the 2s state.
- In the plots of reduced radial functions
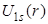 , , 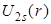 and and 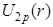 , note the differences in these functions rise from 0. The functions , note the differences in these functions rise from 0. The functions 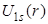 and and 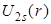 rise like r, in which rise like r, in which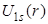 is faster than is faster than 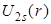 while while 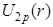 rises like rises like 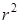 . .
|