Let us start with ground state 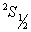 of hydrogen atom having single electron. Here, L =0, S = ½, and J = ½. Under the magnetic field this level splits into two energy levels, essentially due to the spin projection ms = ½ and ms = - ½ because mL = 0. of hydrogen atom having single electron. Here, L =0, S = ½, and J = ½. Under the magnetic field this level splits into two energy levels, essentially due to the spin projection ms = ½ and ms = - ½ because mL = 0.
The splitting energy due to the magnetic field is 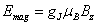 as shown in figure 23.1. The value of gJ = 2. as shown in figure 23.1. The value of gJ = 2.
The transition between this level is governed by the magnetic dipole selection rule
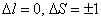 . This transition changes the spin projection by . This transition changes the spin projection by 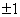 . . |