The best single electron atomic wave functions are the self consistent field (SCF) functions introduced by Hartree (1927).
To obtain these, we start with a set of 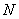 approximate single electron functions. Using of these except the ith function, the average field due to their charge clouds is calculated. approximate single electron functions. Using of these except the ith function, the average field due to their charge clouds is calculated.
This constitutes the potential field in which the ith electron moves. Solution of the ith equation gives an improved wave function 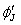 . .
This is repeated for each of the 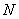 electrons, giving first improved, single electron wave functions. electrons, giving first improved, single electron wave functions.
These are then used to calculate a set of second improved wave functions, and so on; the process is continued until no further improvement is registered.
In other words, the electrons are supposed to move in a potential which they themselves generate. This is the reason it is known as Self Consistent
The calculated energy of the atom is the sum of the energies 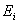 of the individual electrons, minus the coulomb repulsion energy averaged over all pairs of electron wave functions, since this gets included twice over for each pair of electrons of the individual electrons, minus the coulomb repulsion energy averaged over all pairs of electron wave functions, since this gets included twice over for each pair of electrons 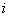 & & 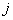 , once in calculation , once in calculation 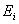 and once in and once in 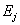 . The energy of each of the SCF/AO’s, is found to be a function of . The energy of each of the SCF/AO’s, is found to be a function of 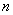 and and 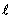 . .
When the central field orbital is multiplied by a spin function 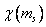 which is which is 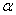 or or 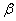 , we have a one-electron, central field spin orbital. , we have a one-electron, central field spin orbital.
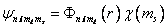 Characterized by the four quantum nos. Characterized by the four quantum nos. 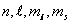 . .
Finally the spin orbitals may be organized into an antisymmetric 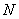 -electron wavefunction in order to satisfy the requirements of the Pauli principle. -electron wavefunction in order to satisfy the requirements of the Pauli principle.

in which 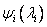 are spin orbitals. are spin orbitals.
The distribution of electrons with 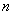 and and 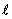 are known as configuration. Electrons in orbitals with same are known as configuration. Electrons in orbitals with same 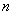 and and  are said to be equivalent. are said to be equivalent. |